





Poster: DDL 2022 (Mitchell et. al)
Flow Rate-Rise Time Profiles from Model Dry Powder Inhaler (DPI) Testing of Abbreviated Impactor Systems Compared with Their Full …
查看文章

Poster: DDL 2022
Developing Methods for Automated Delivered Dose Uniformity (DDU) Testing for Nasal Sprays The COVID-19 pandemic has further stimul…
查看文章

Poster: DDL 2022
Investigating the Impact of Electrostatics on Delivered Dose Uniformity for pMDIs Benchtop in vitro testing is vital to characteri…
查看文章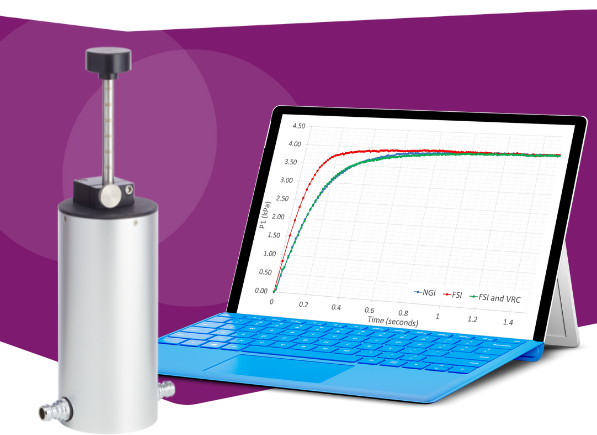

Poster: DDL 2022 (VRC)
Matching Impactor Rise-Time Profiles with a Volume and Resistance Compensator Abbreviated Impactor Measurement (AIM) has several p…
查看文章

DDD 2021 Respiratory eBook Profile
Semi-Automating Inhaler Testing – A Good Idea, but Where to Start? Cascade impaction is a vital technique in the development…
查看文章

ONdrugDelivery Article: November 2020
Semi-Automation in Inhaler Testing – Exploring the Potential and Practicalities In this article, João Pereira, Team Leader R&am…
查看文章
Q&A: OINDPnews 2022
Reducing Inhaler Testing Variability Through Test Environment Control In this Q&A with OINDPnews, our CEO Mark Copley answers …
查看文章

Innovations in Pharmaceutical Technology Article (Samedan): December 2015
Testing Inhaled Generics New product-specific FDA guidance and USP monographs support the development of popular inhaled products….
查看文章

Poster: RDD 2022 (Myatt et al)
The Combined Effect of Valve and Spray Orifice Diameters on pMDI Plume Force Ben Myatt, Barzin Gavtash, Lee Hodges & Andy Coop…
查看文章

Inhalation Article 2012
Measuring the cold freon effect – in vitro testing that can support the development of new and generic metered dose inhalers…
查看文章

Inhalation Article 2012
From actuation to deposition: Particle sizing techniques for characterizing nasal drug delivery systems. An examination of two com…
查看文章

