





Drug Development & Delivery Article: April 2018
Optimizing the Application of In Vitro Test Methods for the Demonstration of Bioequivalence in Orally Inhaled Products
查看文章

Poster: DDL 2022 (Mitchell et. al)
Flow Rate-Rise Time Profiles from Model Dry Powder Inhaler (DPI) Testing of Abbreviated Impactor Systems Compared with Their Full …
查看文章



Inhalation Article 2012
Measuring the cold freon effect – in vitro testing that can support the development of new and generic metered dose inhalers…
查看文章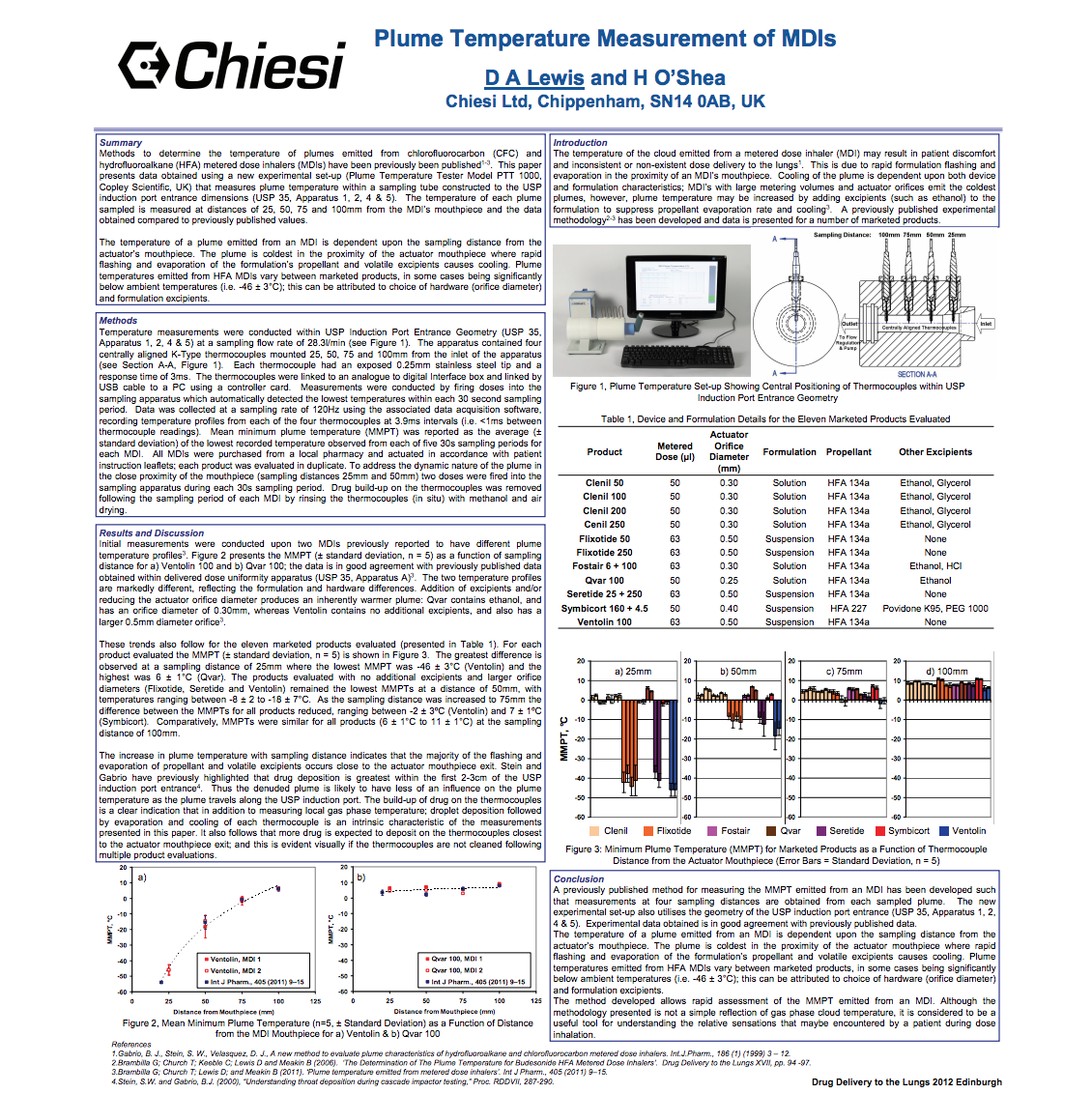



Inhalation Article 2012
From actuation to deposition: Particle sizing techniques for characterizing nasal drug delivery systems. An examination of two com…
查看文章

Inhalation Article 2011
Evaluating the Alberta Throat: An innovation to support the acquisition of more clinically applicable aerosol aerodynamic particle…
查看文章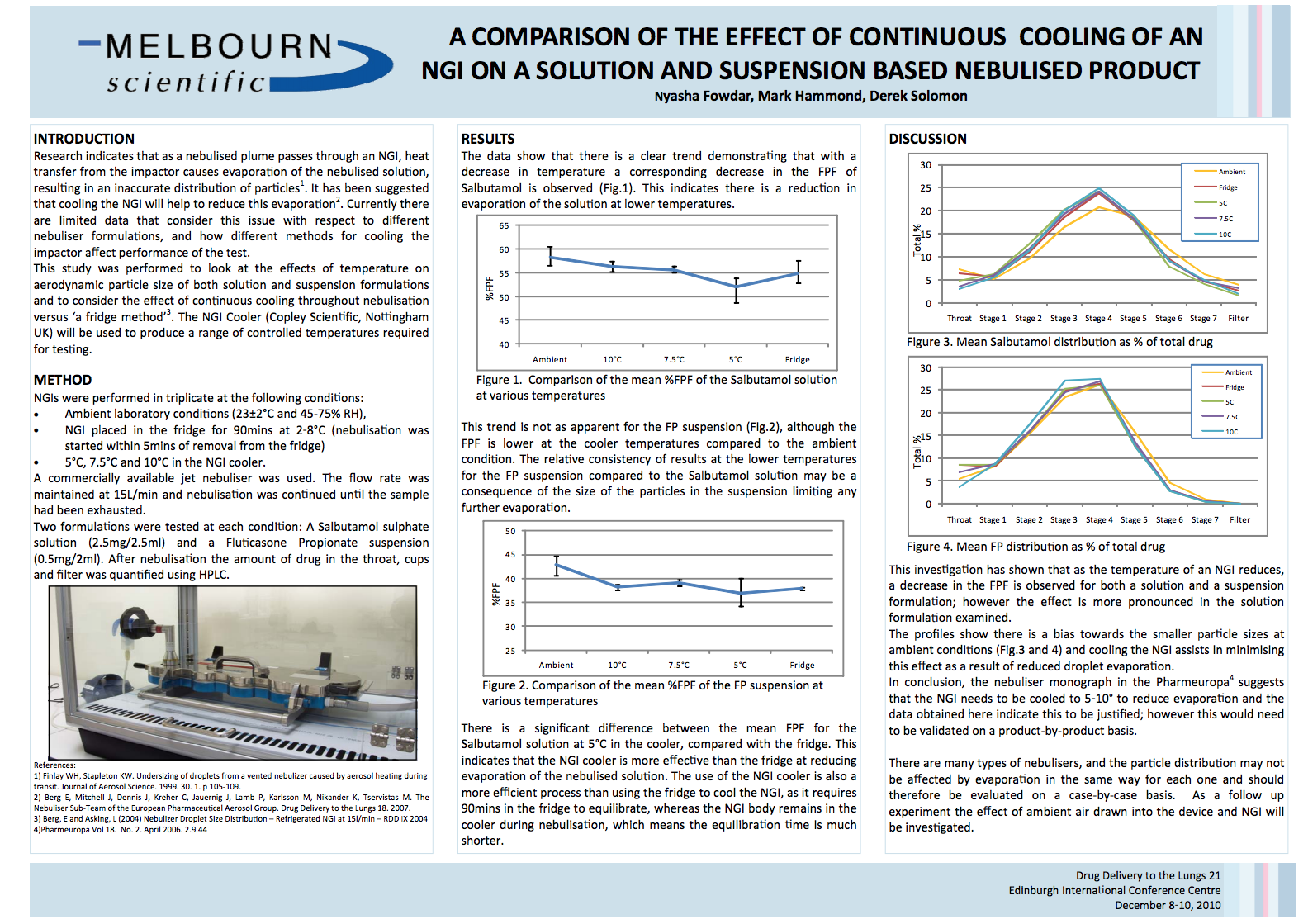

DDL Poster 2010
This paper explains the comparison of the effect of continuous cooling of an NGI on a solution and suspension based nebulised prod…
查看文章

Poster: DDL 2022
Developing Methods for Automated Delivered Dose Uniformity (DDU) Testing for Nasal Sprays The COVID-19 pandemic has further stimul…
查看文章



Poster: DDL 2022
Investigating the Impact of Electrostatics on Delivered Dose Uniformity for pMDIs Benchtop in vitro testing is vital to characteri…
查看文章

