11 November 2014; Nottingham, UK: Copley Scientific will be displaying the smart new Facemask Testing Apparatus for metered-dose inhaler (MDI) testing, in accordance with a recently released draft US Pharmacopoeia (USP) chapter, at Drug Delivery to the Lungs 25 (DDL 25). DDL 25 takes place in the Edinburgh International Conference Centre, Scotland, from 10th to 12th December 2014.
Also on display will be other items from the Copley product range that improve the effectiveness and efficiency of inhaled product testing, including the new NGI Component Carrying and Rinsing Rack, which streamlines post-analysis workflow.
MDIs provide inexpensive and effective relief from asthma and other pulmonary diseases but can be difficult for certain patient groups to use, most especially children and the elderly. The use of spacers and valved holding chambers (VHCs) eliminates the need to coordinate inhalation with actuation of the device, making MDIs easier to operate successfully. Draft USP chapter <1602>, which was released at the beginning of 2014, specifically addresses the testing of MDIs with spacers and VHCs and calls for the testing of facemasks, where they are used, to reflect clinical practice.
The new Facemask Testing Apparatus from Copley Scientific comprises a range of angle-adjustable face models with replaceable skins, a slide rail mechanism and integrated load cell for precise, calibrated control of the applied force when advancing the facemask against the face model. All components are mounted on a base plate using quick release fittings and the apparatus as a whole interface, using the appropriate filter holder and adapters, to the Copley range of breath simulators. The apparatus is also designed to accommodate a wide range of spacer and VHC designs.
The face models used (baby, child and adult variants available) are based on the internationally recognised Laerdal geometries, but the system can be purchased and adapted, by the user, for use with other face models if required. Significant consideration has been given both to the relevance and ease of testing and Copley Scientific is looking forward to gathering initial customer feedback at DDL.
Visit Copley Scientific at DDL 25, 10th to 12th December. Alternatively, log on to www.copleyscientific.com
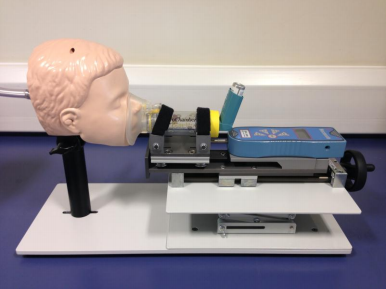
CAPTION: The new Facemask Apparatus from Copley Scientific enables efficient MDI testing in accordance with recently released draft US Pharmacopoeia (USP) chapter <1602>
About Copley Scientific
Copley Scientific is recognised as the world’s leading manufacturer and supplier of inhaler test equipment and is a major provider of testing systems for other pharmaceutical dosage forms. The company is also active in detergent testing, air sampling and aerosol characterisation.
Copley Scientific’s pharmaceutical product range includes test equipment for delivered dose uniformity and aerodynamic particle size measurement of metered-dose inhalers, dry powder inhalers, nebulizers and nasal sprays; as well as tablets (dissolution, disintegration, friability and hardness) capsules, powders, suppositories and transdermals.
Copley Scientific has offices in the UK and Switzerland. The company works in partnership with aerosol particle science experts MSP Corporation in North America and also distributes MSP Corporation’s air sampling and aerosol characterisation products throughout Europe.
Serving the pharmaceutical and associated industries, Copley Scientific offers an extensive range of equipment for research, development and quality control, as well as full validation and aftersales services. This broad range of products is supplied and supported worldwide through a network of specialist distributors.







