Welcome back to our three-part series on the importance of improving in vitro in vivo correlation (IVIVC) in the testing of orally inhaled and nasal drug products (OINDPs).
In part one, we explored why clinical relevance matters and the associated use of throat and nose models. In part two we looked at the application of representative breathing profiles. In this last part we’re looking beyond patient physiology to the trickier issue of patient behaviour.
Why Patient Behaviour Matters in OINDP Testing
Although OINDPs are precision devices, their performance is heavily influenced by patient behaviour and practice. Unlike with tablets or injectables, successful dose delivery with an inhaler depends on the user’s coordination, inhalation technique, and how they handle the device.
If you’ve ever watched someone use an inhaler, or done so yourself, then you’ll be aware that it’s not a simple task and there’s plenty of scope for patient-related variability. Patient behaviour is highly individual with each patient bringing a unique combination of understanding, visual acuity, motivation, dexterity, and capability to their inhaler, with the expectation of consistent well-defined efficacy. The result is a defining challenge for OINDP developers: how do we design products that ensure reliable clinical outcomes, despite the highly significant issue of patient variability?
Real-World Inhaler Use: A Day in the Life of an OINDP
After 10 to 15 years of development, a newly commercialised OINDP finally reaches patients; only to face the challenging realities of everyday misuse.
- The tell-tale crack of an inhaler dropped in a sports bag.
- Forgotten or mis-remembered instructions, like breathing out fully before use.
- A sudden temperature shock when an inhaler comes in from the cold to a warm environment, prior to use.
Out in the real-world, OINDPs unavoidably encounter conditions that diverge substantially from the lab environment. For instance, it’s crucial to be able to rely on an inhaler when travelling to hot and humid climates, or scaling a mountain, as temperature and air pressure drop.
In addition, patients rarely treasure their inhalers despite their life-enhancing role.
To robustly investigate the resulting impacts, we need in vitro methods that not only simulate patient physiology, but also likely patient behaviour. Testing that achieves this aim can provide the information needed to design out the potential for patient-related performance variability.
Regulatory Requirements for Robust OINDP Performance
Reflecting the need to ensure robust OINDP performance, both FDA and EMA guidance recommend a range of drug characterisation studies. Depending on the specific OIP these involve assessing:
- Shaking requirements – especially for suspension-based MDI formulations. Shaking is intended to homogenise the formulation so that each actuation delivers a consistent and accurate dose of drug. Ill-defined or inadequate shaking can lead to variability in the delivered dose.
- Initial priming and re-priming requirements – following storage in various orientations, metering valves may empty/drain, potentially making subsequent actuations less effective. Testing confirms that priming and re-priming instructions correspond with the delivery of a consistent dose.
- Low temperature performance – storage at subzero temperatures can affect the properties of the formulation. Notably, for MDIs, cold conditions may alter propellant evaporation rates, impacting aerosol generation and dose dispersion.
- Performance after temperature cycling – devices may experience repeated temperature fluctuations between subzero and above-room-temperature conditions. This can affect the distribution of formulation within the device and by extension dose delivery.
- Effect of environmental moisture – particularly for DPIs, which as powder formulations are often susceptible to moisture. Exposure to humidity can cause powder clumping or aggregation, altering the delivered dose and its particle size distribution.
Focusing on Robustness
In addition to these drug characterisation studies, there is a specific regulatory requirement to assess and confirm OIP ‘robustness’ the requirement being ‘that the MDI or DPI product is of sufficiently robust design to withstand shipping conditions and typical patient usage.’ Typical patient usage is a somewhat open-ended term, but recently updated EMA guidance offers valuable detail in this area, highlighting the value of:
- Assessing the performance of the device when activated at the frequency indicated in the product information
- Demonstrating the robust performance of any lock-out device or digital sensor
- Vibrational stability studies, especially for powder mixtures
- Drop simulations from heights and orientations that are representative of real-world handling with products that are towards the end of life (e.g. at dose 180 for a 200-dose product) the primary concern being that progressively accumulated drug will be released all at once, delivering a single large dose.

The Drop Test Apparatus DTA enables repeatable and reproducible drop-testing in line with regulatory requirements for robustness assessment.
To be sure of covering all impacts arising from typical patient usage it is also helpful to have a clear understanding of the prevalence of different errors associated with inhaler technique.
Variability in Patient Technique
Studies carried out in clinical settings help to quantify the level of variability in patient technique and the most common area of malpractice. Evidence suggests that the frequency of inhaler technique errors is high with studies showing that 50-80% of patients use their inhaler incorrectly, whether it’s an MDI or a DPI i, ii, iii. Two of the most common errors are:
- Failing to fully exhale prior to inhaler use (up to 80% of patients) iii
- Exhaling into the inhaler rather than away (between 14 and 22% of unsupervised patients)
These mistakes are particularly problematic for dry powder inhalers (DPIs), which rely entirely on patient inhalation for effective drug delivery. While training can improve technique, evidence indicates that errors can be stubbornly persistent, and rigorous or repeated training can be costly.
Some are turning to connected and digital devices as an alternative solution, but here too costs are prohibitive for many healthcare systems. The preferable approach is to develop OINDPs that are inherently less sensitive to this type of misuse. To do this we need to investigate and quantify the extent of a likely impact through targeted, formulation-specific testing.
Case Study: Exhalation into the Device.
Holmes et al iv investigated the impact of exhalation into a Diskus™ DPI (salmeterol/fluticasone 50 µg/250 µg) prior to inhalation. With the aim of generating clinically relevant data the study varied:
- Air flow rate (30, 60, 90 and 120 L/min)
- Distance between mouthpiece and air source (0, 5 and 10 cm)
- Duration (2,4 and 6 s)
- The relative humidity of the air used (28 and 80%)
Key findings:
- Exhalation into the device reduced the delivered dose under all conditions
- At 80% relative humidity, the delivered dose fell below 50% of label claim; drug delivery also became more variable and unpredictable.
- Exhalation into the device at 60 L/min for a duration of 4 s at 5 cm (i.e. conditions around the mid-range of the clinically relevant boundaries set), decreased fine particle fraction (FPF) by ~40% (relative to data measured with no exhalation into the device). This result is attributed to moisture-induced powder agglomeration.
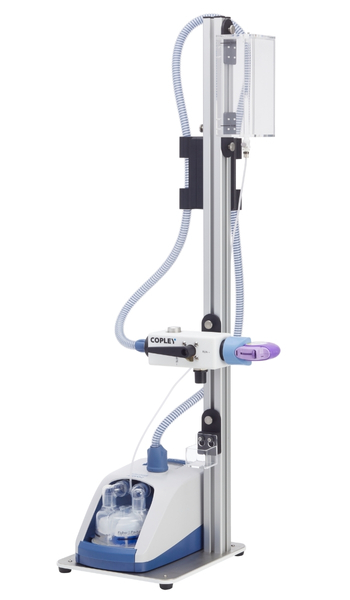
The Patient Exhalation Simulator PES simplifies investigation of the impact of exhaling into a device during product use, a common error in typical patient usage.
These results illustrate how routine patient behaviour can significantly alter inhaler performance, emphasising the importance of refining OINDP design to ensure consistent therapeutic outcomes. For a cohort of asthma patients for which exhalation into the device is ‘typical patient usage’ they suggest that inhalation of a dose well-below label claim will be common. If this noticeably reduces clinical efficacy, then the patient may attempt a further dose, increasing the risk of side effects for a comparable outcome. If it is not noticeable, then there is a greater likelihood of poor disease control over the long term which can mean reduced quality of life and a greater risk of emergency events.
Designing Robust OINDPs for Real-World Use
Designing OINDPs that work well for everyone is critical as we work towards cost-effective treatment for the growing numbers of patients that live with respiratory illness. To achieve consistent efficacy, developers should integrate realistic patient use scenarios into OINDP testing, to:
- Simulate common inhalation technique errors
- Evaluate performance under varying temperature, humidity, and mechanical stress
- Incorporate findings into formulation and device design to minimise variability
Such strategies support the development of OINDPs that deliver reliable, predictable doses across the intended patient population.
Across this series, we’ve examined testing strategies that support the goal of improving the clinical relevance of OINDP testing by revealing and quantifying the impact of patient physiology, inhalation technique and behaviour. Patient behaviour is particularly challenging to address, but an increasing range of tools and methodologies now allow developers to carry out realistic in vitro testing that accounts for real-world variability. By integrating the resulting insights with effective design and formulation principles, it is possible to develop inherently robust OINDPs that are less sensitive to patient technique, ultimately progressing towards more consistent efficacy, minimal side effects and improved patient outcomes.
i J. A. M. Westerik et al ‘Characteristics of patients making serious inhaler errors with a dry powder inhaler and association with asthma-related events in a primary care setting’ J Asthma 2016 53(3):321-329
ii C. L. C. Pessôa et al ‘Most frequent errors in inhalation technique of patients with asthma treated at a tertiary care hospital’ Einstein (Sao Paulo) 2019; 17(2):eAO4397
iii Press, V.G. et al. ‘Misuse of respiratory inhalers in hospitalized patients with asthma or COPD’ Journal of General Internal Medicine 26 (6) 635-42. Jun 2011
iv Holmes, M.S et al ‘An Acoustic-Based Method to Detect and Quantify the Effect of Exhalation into A Dry Powder Inhaler‘. Journal of Aerosol Medicine and Pulmonary Drug Delivery 28(4) 2015
Subscribe now for ongoing expert analysis and technical updates in OINDP testing advancements.








