Everything you need to know to establish a robust baseline for MDI testing, with guidance on extending methods for add-on devices and bioequivalence.
Today we’re beginning a new series of device-focused blogs. Over the course of this year, we’ll shine a spotlight on each class of orally inhaled and nasal drug product (OINDP): metered dose inhalers (MDIs), dry powder inhalers (DPIs), nebulisers/soft mist inhalers (SMIs) and nasal drug products. Each post will provide a clear, practical primer on how to test these devices effectively.
In this first blog, we focus on MDIs. Small, convenient and cost-effective, MDIs remain a frontline defence against respiratory illness. In a recent survey across 53 countries, they accounted for 3 in every 4 inhalers prescribed [i]. While environmental concerns about MDI propellants are growing, their widespread use makes robust and relevant performance testing essential for many analysts.
Whether you’re innovating, reformulating for lower environmental impact, or developing a generic product, DDU (Delivered Dose Uniformity) testing and APSD (Aerodynamic Particle Size Distribution) measurement are key requirements. Together they confirm consistent delivery of a defined respirable dose, the cornerstone of a reliable therapeutic effect.
Understanding How MDIs Work
OINDP testing requirements are shaped by the device operating mechanism and associated patient practice. For that reason, it is helpful to briefly review how MDIs work before looking in detail at the methods.
MDI formulations are solutions or suspensions of an active drug in a propellant or propellant/co-solvent mixture. Surfactants, dispersants and other active ingredients may also form part of the mix. The formulation is held under pressure in a cannister which integrates with the MDI device. Pressing down on the cannister releases a defined dose through the metering valve, dropping the pressure from cannister to atmospheric. This triggers rapid expansion of the propellant, driving dispersion of the formulation into an aerosol.
Key points to note from this are:
- MDIs are ‘active’ devices. The energy for dose delivery and dispersion comes from the formulation, specifically the evaporating propellant.
- MDI formulations can separate over time, for example through settling or creaming of a suspended drug.
- Consistent operation of the metering valve, including filling/refilling of the metering valve chamber (‘priming’), ensures a defined dose.
The patient’s inhalation manoeuvre is relatively unimportant, providing inhalation and actuation are coordinated. Device handling, however, is important as it can affect the homogeneity of the formulation and filling/emptying of the metering valve.
The Basics: Compendial Methods for MDIs
Compendial methods provide an assessment of MDI performance under well-controlled conditions. Collectively they verify that:
- Each actuation delivers a consistent mass of drug: DDU
- Performance is maintained across the lifetime of the product: Delivered Dose Uniformity Over the Entire Contents.
- Aerosolised particles are within the target size range for lung deposition: APSD.
As well as characterising these critical quality attributes for the product, core compendial methods establish a robust reference point. They provide a baseline for evaluating more complex scenarios, such as use with add-on devices or testing across different patient populations.
Delivered Dose Uniformity (DDU) Testing of MDIs

MDI DDU test-set up showing the vacuum pump, flow controller and flow rate sensor used to establish and verify test flow conditions, alongside the dose collection device.
For MDIs, DDU testing is performed at a constant flow rate of 28.3 or 30 L/min, depending on the impactor being used for associated APSD testing. There is no compendial requirement to test with different breathing profiles, which reflects the view that dose delivery is considered independent of the patient’s inhalation manouvre.
Further details of DDU testing requirements can be found in our brochure. In simple terms, however, demonstrating parity across ten sample products is the baseline for confirming batch quality. DDU data confirm that products consistently deliver a defined dose to the patient.
Delivered Dose Uniformity Over the Entire Contents
Alongside product-to-product variability, the multidose nature of MDIs also introduces the potential for variability across the life of an individual inhaler.
Measuring delivered dose at the beginning, middle and end of product life helps assess this, with intervening doses fired to waste under representative conditions. Differences between regulatory authorities and pharmacopoeias add complexity to this type of testing, but we have detailed information to help.
Through-life DDU assessments provide valuable reassurance that an MDI continues to deliver consistent doses from the first actuation to the last. These data are especially helpful for confirming that product use instructions (such as shaking before actuation) are sufficient to ensure the formulation is properly re-dispersed each time the inhaler is used.
If the formulation is not adequately dispersed prior to actuation, this may be reflected in variability between successive doses, or a progressive drift in delivered dose per actuation over the lifetime of the product. This is particularly likely with drugs prone to settling or creaming during storage.
Aerodynamic Particle Size Distribution (APSD) Measurement of MDIs

MDI APSD measurement set-up, showing the vacuum pump, flow controller and flow rate sensor to establish test conditions, and a Next Generation Impactor (NGI) for size fractionation of the dose.
Cascade impaction provides an APSD for the sub-10 micron fraction of the delivered aerosol. The focus measurement is on the fraction below 5 microns in aerodynamic diameter. Based on size, this fraction is most likely to deposit in the lungs. DDU and APSD are therefore complementary: the first revealing how much drug is delivered, the second indicating how much of that delivered dose is likely to reach its target – the lungs.
APSD measurement for MDIs is also performed under constant flow rate conditions. Depending on the cascade impactor used, the specified flow rate is either 28.3 L/min or 30 L/min. Details on establishing a cascade impactor test set-up for MDIs and on test conditions are covered in one of our earlier blogs.
MDIs with Add-on Devices
Add-on devices, such as spacers and valved holding chambers (VHCs), help make MDIs accessible to patients who struggle to coordinate inhalation with actuation, for example young children.
Both introduce volume between the inhaler and the patient, decoupling actuation from inhalation. VHCs also incorporate a one-way valve that retains the aerosolised dose, allowing patients time to take a slow deep breath after actuation and preventing exhalation back into the device.
As add-on devices can alter the characteristics of the delivered dose, separate compendial methods are defined for testing MDIs in combination with these accessories.
Delivered Dose Uniformity (DDU) Testing of MDIs with Add-on Devices
Compendial DDU methods for MDIs with add-on devices specify testing under tidal flow conditions to better reflect patient use. The associated test set-ups are also useful for those wishing to establish better in vitro-in vivo correlations (IVIVCs) for MDIs.
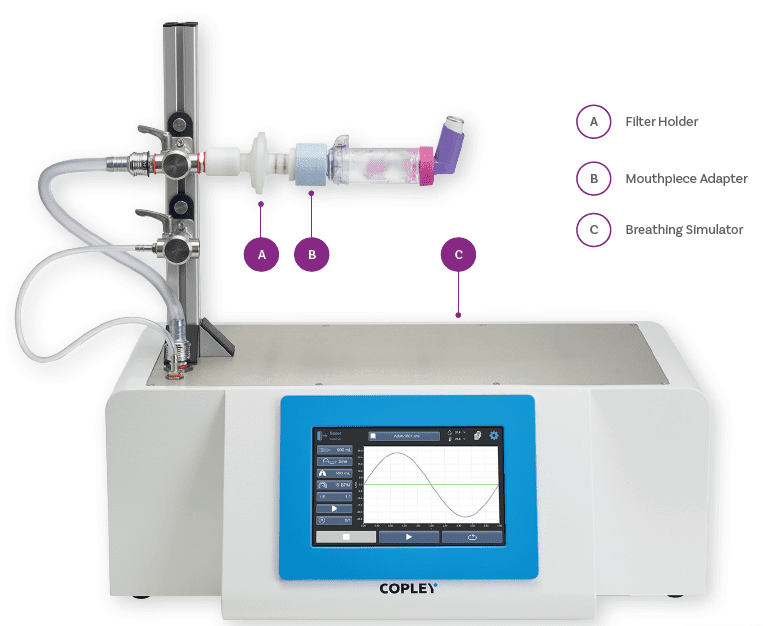
MDI DDU test set-up incorporating a breathing simulator for testing with add-on devices under controlled tidal breathing conditions.
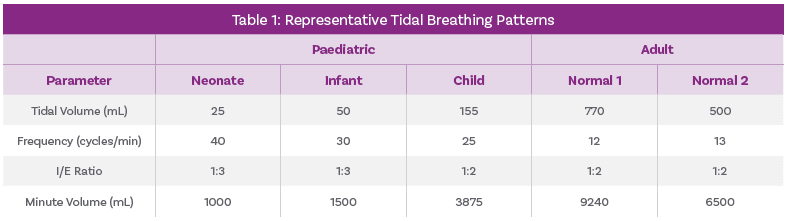
Representative tidal breathing pattern parameters used for DDU testing of MDIs with add-on devices.
Aerodynamic Particle Size Distribution (APSD) Measurement of MDIs with Add-on Devices
Compendial methods for APSD measurement for MDIs with add-on devices capture the impact of:
- Incorporating the add-on device.
- Introducing a delay between actuation and inhalation (VHC only).
This second scenario often has the greatest impact on dose characteristics. Residence time in the VHC allows for continuing propellant evaporation and/or droplet agglomeration, potentially altering particle size.
Standard test set-ups for APSD measurement for MDIs, as shown earlier, enable all necessary testing.
Regulatory Requirements for the Demonstration of Bioequivalence (BE)
While compendial DDU and APSD methods establish baseline for MDI performance, regulatory guidance increasingly calls for more clinically realistic in vitro testing to support bioequivalence submissions. More specifically, FDA Product Specific Guidances (PSGs) for MDIs recommend Realistic APSD measurements and dissolution testing.
Realistic APSD measurements typically involves using mouth-throat models of different sizes and representative breathing profiles that together represent the range of patients who may use the product. In practice this means replacing the standard USP induction port with more clinically realistic mouth-throat models and moving beyond constant flow rate test conditions.
By combining compendial methods with these more patient-representative approaches, developers can build a stronger case for equivalence between test and reference products across the patient population, supporting regulatory approval while minimising reliance on clinical trials.
A full discussion of requirements for dissolution testing lies beyond this blog but you’ll find plenty of information and practical solutions here.
Arguments for Automation
Automation plays an important role in ensuring consistent, high-quality data across both DDU testing and APSD measurement. For example, automated shake and fire systems standardise key handling and actuation actions that are most likely to introduce inconsistency including:
- Shaking technique
- Delay time between shaking and firing
- Actuation force profile
- Length of time between repeat firings
- Storage orientation during storage
The result is more reliable and repeatable dose delivery and dispersion, supporting improved data quality.
Automation also reduces the burden of firing MDIs to waste. Since fire to waste must be performed under conditions that replicate product use, automation supports data integrity, while also improving analyst productivity (and job satisfaction).

Vertus®+ III, an automated shake and fire system for MDIs for DDU testing and APSD measurement
MDI testing: a summary
Core compendial methods for MDIs – DDU testing and APSD measurement – are relatively straightforward. More sophisticated strategies are required when:
- Assessing the impact of add-on devices
- Measuring Realistic APSD to support BE.
Constant flow rate | Patient representative flow profiles | USP induction port | Mouth-throat models | |
DDU Compendial methods – MDI only | ||||
APSD Compendial methods – MDI only | ||||
DDU Compendial methods – MDI + add-on device | ||||
APSD Compendial methods – MDI + add-on devices | ||||
Realistic APSD/ Better IVIVCs |
Across all testing scenarios, automating actuation is valuable because of the multiple ways in which MDI storage, handling and actuation can influence product performance.
[i] IPCRG. (2024). An assessment of Pressurized Metered-Dose Inhaler use in countries in Europe and the rest of the world | IPCRG. [online] Available at: https://www.ipcrg.org/24041.
Next in the series:
We explore testing for dry powder inhalers, where flow-dependence introduces a different set of challenges. For a deeper dive into the topics covered here, check out our other blogs on cascade impactor setup, MDI reformulation, and the benefits of automation.








