Everything you need to know to establish a robust baseline for DPI testing, including robust flow control, with guidance on extending methods to more clinically realistic conditions for bioequivalence.
Welcome to the second in our series of device-focused blogs. This post provides a clear, practical primer on how to test dry powder inhalers (DPIs), covering core compendial methods for Delivered Dose Uniformity (DDU) testing and Aerodynamic Particle Size Distribution (APSD) measurement, as well as additional considerations relevant to product development and bioequivalence studies.
In our first blog in this series, we focussed on metered dose inhalers (MDIs). Today we’re turning to DPIs. Propellant-free by design, DPIs have an inherently low carbon footprint that can help to reduce the environmental impact of effective respiratory healthcare. Many patients also find DPIs easy to use because there is no need to coordinate inhalation with device actuation.
Such benefits drive ongoing interest in DPI technology which is also widely used when higher drug loadings are required, including for systemic action.
Whether you’re innovating new DPI devices, optimising formulation characteristics or developing a generic product, DDU testing and APSD measurement are key requirements. Together they confirm that a consistent respirable dose is delivered to the patient, which underpins reliable therapeutic performance.
A defining characteristic of DPIs is that performance depends strongly on the patient’s inhalation effort. Test methods must therefore reflect the pressure drop and inhalation volume associated with patient use.
In this blog:
- How DPIs work and why this matters for testing
- Compendial methods for DDU and APSD
- Regulatory requirements for demonstrating bioequivalence (BE)
- How inhalation profiles influence DPI performance
Understanding How DPIs Work
A brief review of how DPIs work helps explain why DPI testing methods are designed the way they are.
The defining challenge with DPIs is that particles that are fine enough to reach the lungs typically flow poorly and tend to agglomerate. This complicates device emptying and delivery of a high respirable fraction (particles below 5 µm aerodynamic diameter).
To address this, DPI formulations generally fall into one of two categories: carrier and carrier-free.
In carrier-based formulations, very fine active drug particles are attached to a coarser carrier, such as lactose. The larger carrier particles improve powder flow, handling and metering within the device.
Carrier-free formulations contain loose clusters of fine active drug particles alone, sometimes with very small amounts of active excipient to promote dispersion.
With either type of formulation sophisticated particle engineering techniques such as micronisation, spray drying or other advanced processes may be required to achieve suitable flow and dispersion behaviour.
Patients are instructed to take a fast, forceful, deep breath when using a DPI. This inhalation draws air through the powder formulation, fluidising and dispersing it into an aerosol. The airflow must generate enough energy to empty the device and either separate the drug particles from the carrier or de-agglomerate a carrier-only formulation thereby delivering particles small enough to reach the lungs (a respirable dose).
Key points to note are:
- DPIs are ‘passive’ devices. The energy required for dose delivery and dispersion comes entirely from the patient’s inhalation effort.
- DPI devices are designed to ensure that the pressure drop generated by inhalation produces sufficient turbulence to disperse the drug.
- Differences in device design give rise to differences in internal resistances. As a result, the airflow required to achieve the same pressure drop varies between devices.
These characteristics explain why DPI test methods are based on pressure drop and inhaled volume.
The Basics: Compendial Methods for DPIs
Compendial methods assess DPI performance under conditions that reflect the strength and duration of a typical patient inhalation. They verify critical quality attributes for the product, confirming that:
- Each actuation delivers a consistent mass of drug: DDU
- Performance is maintained across the lifetime of the product: DDU Over the Entire Contents.
- Aerosolised particles fall within the target size range for lung deposition: APSD.
As introduced earlier, together DDU and APSD confirm that the device delivers a consistent respirable dose.
To reflect patient use, compendial test methods are based on generating a defined pressure drop across the device (4kPa), which is representative of the average inhalation strength of a healthy adult. Since each DPI has a different internal resistance, the airflow required to generate this pressure drop varies between devices. During method setup, analysts therefore adjust the airflow until a 4 kPa pressure drop is achieved across the inhaler. As a result, high-resistance devices are tested at lower flow rates, while low-resistance devices require higher flow rates to achieve the same pressure drop.
Delivered Dose Uniformity (DDU) Testing of DPIs
For DPIs, DDU testing conditions are determined for each individual product using the approach described above. During method set-up, analysts establish:
- The air flow rate required to generate a 4 kPa pressure drop across the device (to an upper limit of 100 L/min).
- The test duration to achieve a defined inhaled volume
DPI DDU test-set up showing the vacuum pump, critical flow controller and flow rate sensor used to establish and verify test flow conditions, alongside the dose uniformity sampling apparatus (DUSA) for dose collection.
The total inhaled volume used during testing depends on the pharmacopoeial method:
- 2 L total inhaled volume (USP / FDA)
- 4 L total inhaled volume (Ph. Eur. / EMA)
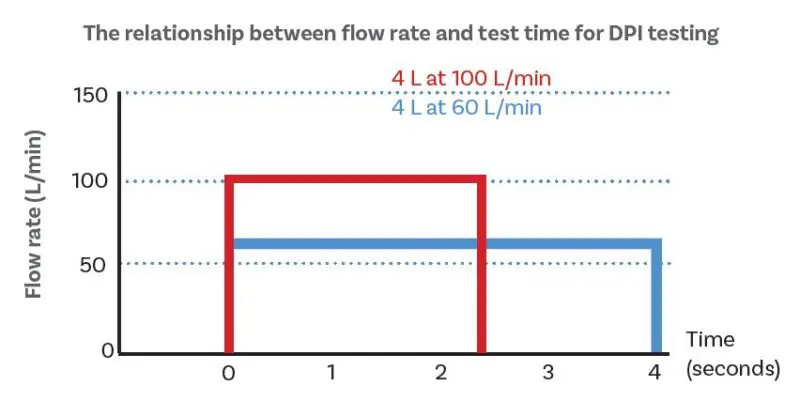
DPIs are tested at the flow rate associated with a 4 kPa pressure drop across the device, with test time based on total inspiration volume.
Once the appropriate flow rate has been established, testing is performed under critical flow conditions to ensure that airflow remains stable throughout the test.
The Critical Flow Condition
Since dose delivery and powder dispersion are highly sensitive to flow rate, critical flow conditions are used to ensure this does not introduce variability..
Critical flow occurs when the pressure downstream of a valve falls below ~50% of the upstream pressure. At this point, air flow through the valve reaches sonic velocity, and so further reductions in downstream pressure do not increase the flow rate.
Air flow rate becomes dependent only on upstream pressure.
The test set-up for DDU testing includes a Critical Flow Controller. This unit incorporates a flow control valve and allows analysts to establish critical flow across it. Air flow through the DPI under test is therefore highly stable. Any downstream pressure fluctuations caused by the vacuum pump have no impact.
For DPIs, critical flow conditions ensure reproducible test conditions, and by extension, consistent dose delivery.
Delivered Dose Uniformity Over the Entire Contents
DPIs can be classified into three main categories:
- single-unit dose devices, typically disposable pre-loaded products or devices into which the patient loads a single capsule before each use.
- multi-unit dose devices, in which pre-metered doses are held in discs or blister packs.
- multi-dose reservoir devices, in which up to 200 doses are held in a bulk formulation reservoir.
For multidose DPIs, there is therefore potential for variability across the life of an individual inhaler, just as with an MDI. Measuring delivered dose at the beginning, middle and end of product life assesses this, and we offer detailed guidance for those navigating specific pharmacopoeial specifications.
Through-life DDU assessments confirm that a DPI continues to deliver consistent doses from the first actuation to the last. With DPIs there is potential for formulation to adhere to the walls of the device which may later detach and lead to unexpectedly large doses. Through-life data detect issues such as this.
Aerodynamic Particle Size Distribution (APSD) Measurement of DPIs
While DDU confirms that a defined dose is delivered, APSD confirms that the aerosol is dispersed to a respirable size.

DPI APSD measurement set-up, showing the vacuum pump, critical flow controller and flow rate sensor used to establish and control test conditions, and a Next Generation Impactor (NGI) with a preseparator for size fractionation of the dose.
APSD is measured using a cascade impactor such as the NGI (as shown) or Andersen Cascade Impactor (ACI). Testing is performed at the same flow rate as DDU testing, under the same critical flow described above.
In addition:
- A preseparator is inserted between the induction port and the impactor to capture large carrier particles and prevent overloading of the early impactor stages.
- Cup coating should be considered to reduce the risk of particle bounce (see box).
- Environmental control may be necessary to minimise the impact of humidity and/or static.
Environmental control is especially relevant for DPIs because of the susceptibility of powders to moisture and electrostatic effects. For this reason, controlling laboratory humidity and minimising electrostatic effects can help improve the repeatability of DPI testing. We have information on this topic, and tools to help.
Cup Coating: What is it and Why is it Used?
Cup coating involves depositing a tacky, viscous layer on the collection surface of each stage of an impactor. Substances such as glycerol or silicone oil are often used.
Cup coating can stop particle bounce. This is when a particle hits the collection surface of the impactor and instead of depositing, bounces off, and re-enters the air flow. This compromises accurate APSD measurement and is most common with hard and/or dry particles.
Please see our brochure for more detailed information about the practicalities of cup coating.
Regulatory Requirements for the Demonstration of Bioequivalence (BE)
While compendial DDU and APSD methods establish the baseline performance for a DPI, regulatory guidance for the demonstration of BE increasingly recommends additional in vitro tests that better reflect patient physiology and provide further information about in vivo fate.
For example, FDA Product Specific Guidances (PSGs) for some of the most common DPIs recommend:
- Realistic APSD (rAPSD) measurements using:
o Anatomically representative mouth-throat models in place of the right-angled induction ports used for compendial testing.
o Breathing profiles that are representative of the target patient population in place of the constant on/off air flow profile used for compendial testing. - Dissolution testing to detect differences in the dissolution behaviour of the delivered dose between Test (T) and Reference (R) products.
- Particle morphology characterisation to detect differences in the particle shape, surface structure and agglomeration state between T and R products.
These approaches allow developers to build a stronger case for BE across the intended patient population, helping to reduce reliance on clinical trials.
More generally, the test set-ups used for rAPSD (see below) also allow researchers to explore the impact of inhalation profiles, a defining factor in DPI performance.

A test set-up for realistic APSD measurement set-up incorporating a breathing simulator for the application of realistic inhalation profiles, an Alberta Idealised Throat for clinically relevant representation of the mouth/throat.
Mixing Inlet: What is it and Why Do I Need One?
A mixing inlet allows analysts to apply variable inhalation profiles across the product under test while maintaining the fixed flow conditions required by a cascade impactor.
Cascade impactors can only operate at a constant flow rate. Exploring the impact of inhalation profile means applying variable flow across the inhaler under test to simulate patient breathing.
The mixing inlet resolves this incompatibility. It decouples air flow through the inhaler from the air flow through the impactor. This allows a variable air flow rate to be applied across the DPI, while the impactor continues to operate at a stable, constant flow.
Mixing inlets are therefore commonly used in studies where patient-representative inhalation profiles are applied.
Exploring the Impact of Inhalation Profile
An important limitation to DPI use is that certain patient groups may not be able to inhale with sufficient strength to achieve effective dose delivery and dispersion. Improving device design and formulation properties to broaden potential application is a key focus for DPI development.
Testing to assess the impact of inhalation profile helps researchers to:
- Identify strategies for improving device design
- Optimise formulation properties
- Ensure robust clinical efficacy across all target patient populations
Using a breathing simulator, analysts can replace the on/off air flow associated with compendial testing with one that is more representative of patient physiology (see below).
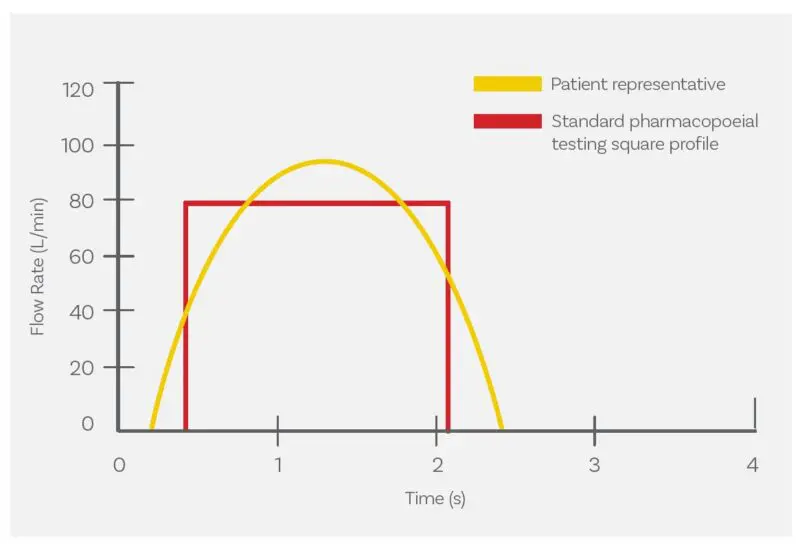
Humans generate a tidal breathing pattern that results in a wave-shaped inhalation profile (yellow) rather than the step-change associated with compendial test set-ups (red).
Two defining characteristics of a patient-representative inhalation profile are:
- Peak inspiratory flow rate (PIFR) – the maximum air flow achieved during inhalation.
- Flow rate rise-time to peak flow – the time taken for air flow to accelerate from zero to a peak value.
There is a growing body of research indicating that the flow rate rise time is especially important for DPI performance. This is likely because it defines conditions in the critical first milliseconds, when powder fluidisation and aerosolisation begin.
With a breathing simulator both flow rate rise time and peak inspiratory flow rate can be independently studied to robustly assess how different inhalation characteristics influence dose delivery and dispersion.
Introducing the VRC: Matching Flow Rate Rise-Times
The figure below shows inhalation profile data for two APSD test set-ups for DPIs, one with an NGI (blue) the other with a Fast Screening Impactor (FSI – red). The test set-ups are otherwise identical. Note: P1 is a proxy variable for flow rate.
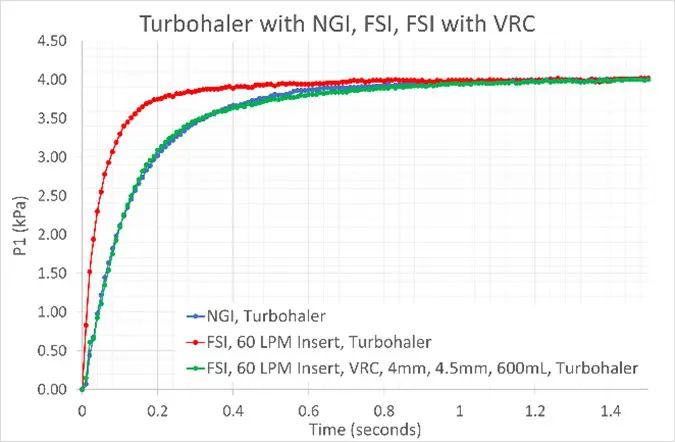
The Volume and Resistance Compensator (VRC) allows analysts to match flow rate rise time across full resolution and abbreviated impactor test set-ups.
The FSI is an apparatus for Abbreviated Impactor Measurement (AIM). [GL12.1]It has just one stage and a much smaller internal volume than the NGI. The result – a much faster flow rate rise-time under otherwise identical conditions.
Studies indicate that this difference can impact APSD measurements. The Volume and Resistance Compensator (VRC) allows analysts to correct for this by independently adjusting:
- Flow resistance
- Test set-up volume
Incorporating a VRC in the FSI test set-up (green line) matches the flow rate rise-time profile to the NGI. This removes an important source of variability in DPI testing, ensuring data reflect product behaviour rather than differences in test set-up.
DPI Testing: A Summary
Unlike MDIs and SMIs, where the device provides the energy for aerosol generation, DPIs rely entirely on the patient’s inhalation effort to empty the device and disperse the powder formulation. As a result, DPI test methods are designed to reflect the pressure drop and inhalation volume associated with patient use.
This leads to several defining features of DPI testing:
- Product-specific flow rates and test duration in compendial DDU and APSD methods
- Critical flow conditions to ensure flow rate stability
- Investigation of inhalation profile, particularly PIFR and rise time, during product development and BE studies
Achieving consistent dose delivery and effective dispersion using solely the inspiratory effort of the patient is a central challenge for DPI products. Robust in vitro testing provides the data needed to optimise device development, refine formulation properties and ensure reliable therapeutic performance across the intended patient population.
This blog provides a practical overview of DPI testing fundamentals, but many of the topics discussed here can be explored in greater depth.
If you are setting up DPI testing or refining existing methods, you may find these resources helpful:
- Guidance on cascade impactor setup
- Practical advice on flow control and pressure drop measurement
- Strategies for environmental control in DPI testing
You can explore these topics and additional practical guidance in our related articles and application notes.
Next in the series:
We’ll explore testing for nebuliser and soft mist inhalers (SMIs), where active aerosol generation introduces a different set of challenges.









