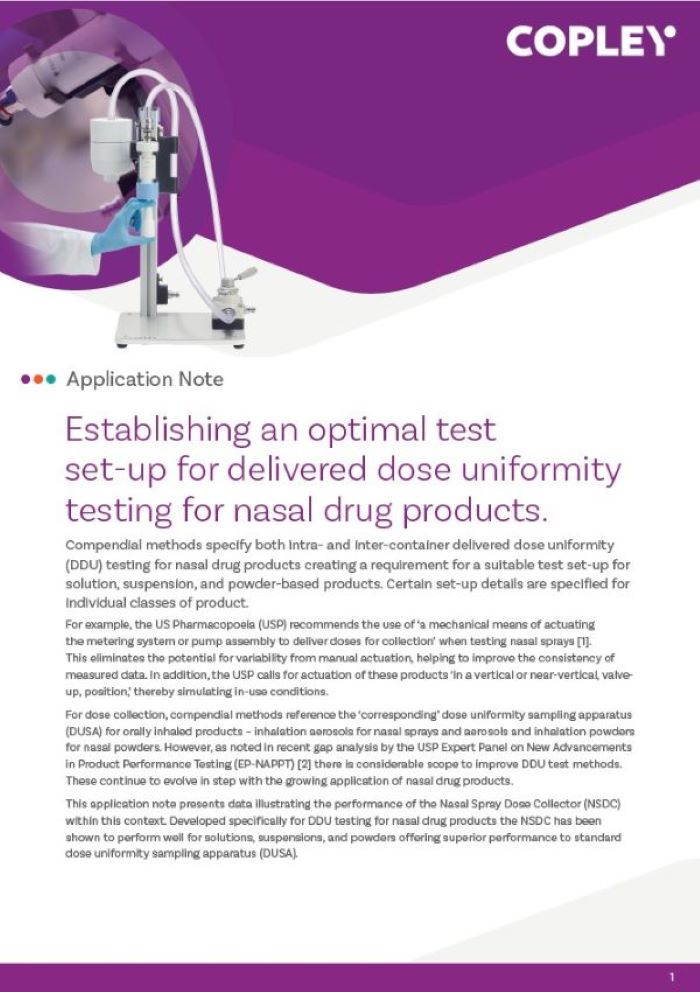
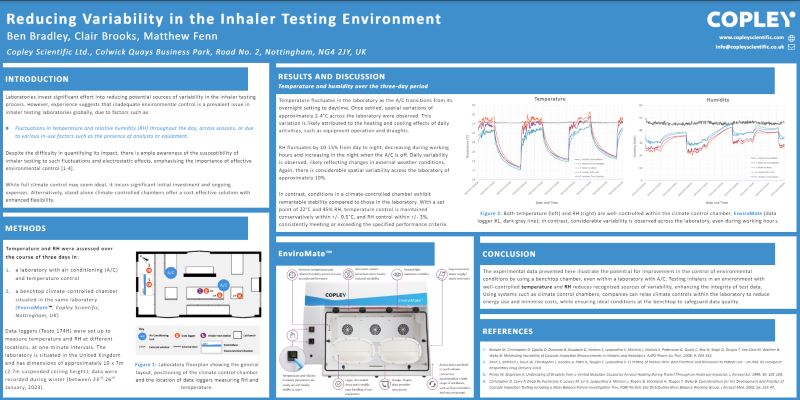
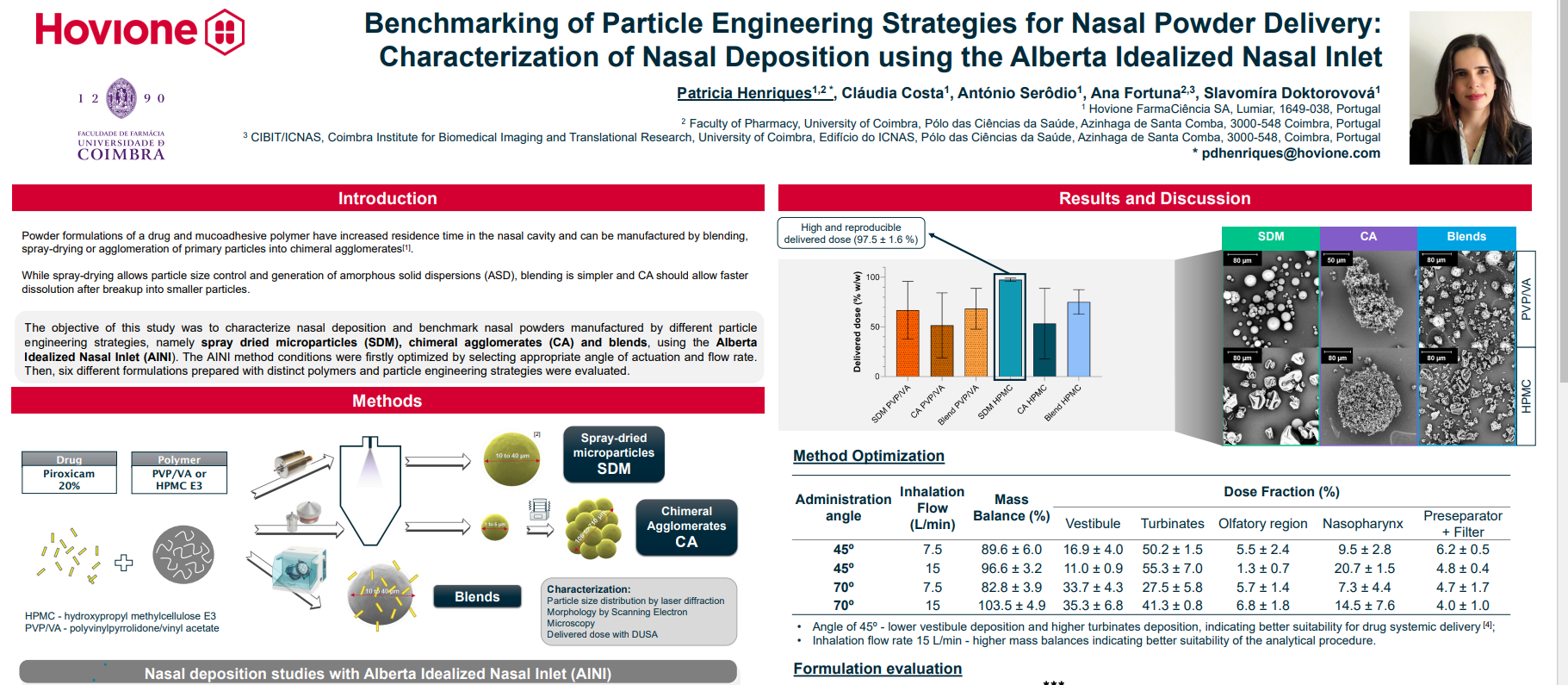
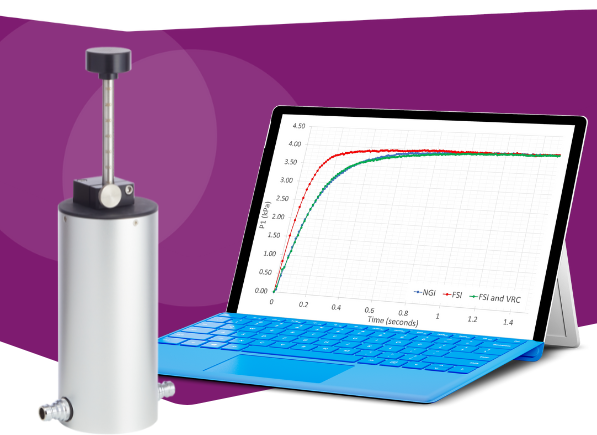
Content Library
Access the latest product brochures, product specifications, whitepapers and more from Copley.


You’ve viewed 15 of 76 posts

Contact Us Today
Have a question? Our friendly and experienced team are here to help.