Contact Us
Contact Us
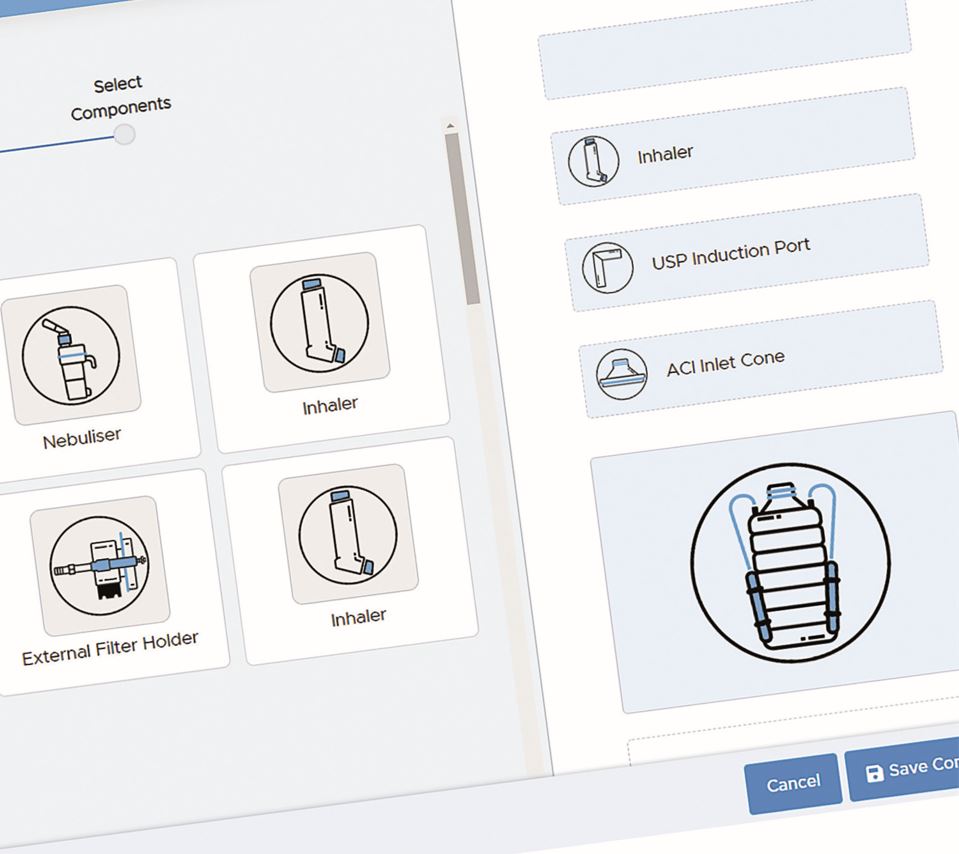
How Does a Cascade Impactor Work?


MDIs: Semi-Automated APSD Measurement
The Vertus® III automated shake, fire and shot waste range is made up of integrated turn-key solutions for precise, controlled and reproducible MDI testing. Compatible with most MDIs, the Vertus III systems offer analysts complete control over:
- The speed, angle and duration of shaking, ahead of actuation
- Firing force and the speed of application and release of that force
- The time delay between the end of shaking and device actuation
Breath-Actuated MDIs (BAIs)
The Breath Actuation Controller BAC 100i can also be used for the testing of Breath-Actuated (or Breath-Operated) MDIs. In this case, the BAC 100i is used to initiate the flow, simultaneously triggering the breath-actuated inhaler.
Automation Tools
Related Services



Training
We offer a range of training courses, presentations and seminars covering a wide range of topics.
Find Out More


Servicing
A comprehensive range of both in-house and on-site product servicing options are available
Find Out More


Support
Our team of experienced technicians and engineers are on hand to help and advise
Find Out More